Rice bran series
Summary
In the 1890s, a Dutch military physician, Christiaan Eijkman and his assistant discovered that polished “white rice” could cause leg weakness on chickens. It was later proved that the Vitamin B1 removed during the polishing (i.e. the rice bran) was the cause of sick chickens[1]. Subsequently, the micronutrients rich in rice bran were continuously discovered, such as oryzanol, sitosterol, vitamin E, squalene, etc.[2]
Since its establishment in 1994, Delekang has been dedicated to research and manufacture of rice bran series products. We have successively developed oryzanol, ferulic acid, rice bran sterol and phytosterol, continuously exploiting the value of rice bran.
History of rice bran series
-
In July 1995, gamma oryzanol was launched in the market.
-
In 2003, Delekang developed the rice bran oil of first-class in national standard.
-
In 2005, ferulic acid was introduced.
Rice bran oil
Introduction
In the perspective of nutrition, oils and fats is the carrier of fat-soluble Vitamin A, D and E, and also an important part of cell walls and other biological structures. In cooking, oils and fats also affect the flavor of food. [3]
Vitamin E, including our tocopherols and four tocotrienols [4], is a fat-soluble antioxidant that protects cell membrane from reactive oxygen species (ROS). [5]
Compared with tocopherols, tocotrienols have stronger anti-oxidant property[6], which are naturally derived from plant oil, especially rice bran oil.[7]
The content of Vitamin E in plant oils(mg/kg)[7]
| Edible plant oil | Tocopherols | Tocotrienols | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Sample size | Concentration(mg/100g) | Sample size | Concentration(mg/100g) | ||||||
| α-T | β-T | γ-T | δ-T | α-T3 | (β+γ)-T3 | δ-T3 | |||
| Coconut oil | 3 | ... | ... | ... | ... | 2 | 2.89 | 1.52 | ... |
| Palm kernel oil | 3 | 0.62 | ... | ... | ... | 2 | 1.94 | ... | ... |
| Sesame oil | 46 | 2.67 | 0.26 | 41.16 | 1.11 | 15 | ... | ... | ... |
| Walnut oil | 2 | 2.71 | 0.17 | 24.12 | 2.58 | - | - | - | - |
| Flaxseed oil | 9 | 3.42 | 1.79 | 36.44 | 1.26 | 1 | 1.00 | ... | ... |
| Palm stearin | 2 | 8.44 | ... | ... | ... | 2 | 7.78 | 11.81 | 2.25 |
| Blend oil | 35 | 10.76 | 1.49 | 57.03 | 17.75 | - | - | - | - |
| Soybean oil | 94 | 11.39 | 2.57 | 65.40 | 23.74 | 31 | 0.05 | 0.03 | 0.04 |
| Tea seed oil | 28 | 12.2 | 0.12 | 0.40 | 0.15 | 1 | ... | ... | ... |
| Canola oil | 1 | 13.12 | ... | 35.69 | 0.54 | - | - | - | - |
| Olive oil | 82 | 14.38 | 0.72 | 1.52 | 0.29 | 30 | 0.48 | ... | ... |
| Camellia oil | 20 | 14.81 | 0.49 | 0.68 | 0.14 | 6 | ... | ... | ... |
| Palm oil | 19 | 15.26 | 0.53 | 2.92 | 1.11 | 5 | 9.47 | 21.65 | 2.63 |
| Grapeseed oil | 4 | 18.98 | 0.79 | 8.22 | 1.23 | 1 | 7.73 | 14.08 | 0.38 |
| Rapeseed oil | 68 | 19.64 | 0.57 | 39.25 | 4.49 | 21 | ... | ... | ... |
| Rice bran oil | 18 | 19.84 | 0.86 | 4.54 | 0.95 | 11 | 12.18 | 15.81 | 0.41 |
| Peanut oil | 71 | 21.68 | 1.38 | 16.40 | 2.00 | 19 | ... | ... | ... |
| Corn oil | 74 | 27.45 | 1.77 | 54.28 | 3.25 | 19 | 0.59 | 4.00 | ... |
| Safflower oil | 4 | 31.82 | 0.52 | 0.50 | 0.07 | - | - | - | - |
| Cottonseed oil | 3 | 55.31 | ... | 40.25 | 2.69 | 3 | 2.82 | ... | ... |
| Sunflower oil | 72 | 62.98 | 3.86 | 3.70 | 0.65 | 72 | ... | ... | ... |
| Wheat germ oil | 1 | 169.57 | 69.28 | 16.77 | 1.09 | - | - | - | - |
Specification
Based on the national standard GB/T 19112-2003
| Items | Class 1 | Class 2 | Class 3 |
|---|---|---|---|
| Color Lovibond 5 1/4″ | Y ≤ 35 R ≤ 3.5 | Y ≤ 35 R ≤ 5.0 | -- |
| Color Lovibond 1″ | -- | -- | Y ≤ 35 R ≤ 3.0 |
| Acid value (mg KOH/g) | ≤ 0.20 | ≤ 0.30 | ≤ 1.0 |
| Peroxide value (mmol/kg) | ≤ 5.0 | ≤ 5.0 | ≤ 7.5 |
| Solvent residue (mg/kg) | ND | ND | ≤ 20 |
| Soap content (%) | -- | -- | ≤ 0.03 |
| Insoluble impurities (%) | ≤ 0.05 | ≤ 0.05 | ≤ 0.05 |
| Moisture and volatiles (%) | ≤ 0.05 | ≤ 0.05 | ≤ 0.10 |
| Smoke point (°C) | ≥ 215 | ≥ 205 | -- |
| Refrigeration test (0°C) | clear and transparent in 24h | clear and transparent in 5.5h | -- |
| Iodine value (g/100g) | 92-115 | 92-115 | 92-115 |
Packaging
| Type | Package |
|---|---|
| Bulk | 190kg/drum、flexitank |
| Retailing | 500mL、1L、1.5L、4L、5L |
Storage
Store in light-tight, airtight and dry condition
Gamma Oryzanol
CAS number: 11042-64-1
HS code: 29189900.90
National standard code: WS1-47(B)-89-2016
Introduction
Gamma oryzanol is a lipid composite mainly derived from rice bran. It is used to improve climacteric syndrome, anxiety, insomnia in Japan,[8] while in China, it is used for dysautonomia premenstrual syndrome and climacteric syndrome. [9]
Structure


β-sitosteryl ferulate (5-10%)

Campesteryl ferulate (10-15%)

Cycloartenyl Ferulate (25-30%)

24-methylene-cycloartanyl ferulate (35-40%)
Specification
| Items | Standard |
|---|---|
| Assay | 95.0 ~ 102.0% |
| Loss on drying | ≤ 1.0% |
| Residue on ignition | ≤ 0.3% |
| Heavy metal | ≤ 20 mg/kg |
| Appearance | White or white-ish, odorless powder |
| Solubility | Soluble in acetone and trichloromethane, slightly soluble in n-heptane and ethanol, insoluble in water |
Packaging
20 kg/drum
Storage
Store in light-tight, airtight and dry condition
Ferulic acid
CAS number: 537-98-4
HS code: 29189900.90
Other name: (2E)-3-(4-hydroxy-3-methoxyphenyl)prop-2-enoic acid
Chemical formula C10H10O4
Molecular mass 194.184
Introduction
Ferulic acid is a very common component in natural plants and usually used as a raw material for spices. It mainly exists in the cell wall. Its tronger antioxidant property can protect cell, which make ferulic acid applicable as antioxidant and photostage inhabitation in cosmetics [10]. Ferulic acid also presents in some herbal medicines, such as female ginseng, Cimicifuga foetida and Ligusticum wallichii. [11]
Structure
Specification
| Items | Standard |
|---|---|
| Content | ≥ 99.0% |
| Loss on drying | ≤ 0.5% |
| Residue on ignition | ≤ 0.2% |
| Heavy metal | ≤ 10 mg/kg |
| Arsenic | ≤ 2 mg/kg |
| Melting point | 170.0~174.0°C |
Packaging
20 kg/drum
Storage
Store in light-tight, airtight and dry condition
Phytosterol
CAS number: 83-46-5
HS number: 29061990.90
Introduction
Sterols are the major trace substance in fat.
Cholesterol is a type of sterols, the main sterols in animal’s fat. There are two types of sterols in human blood: high-density lipoprotein (HDL)cholesterol and low-density lipoprotein (LDL) cholesterol. Excessive accumulation of LDL cholesterol in the serum can lead to blockage of arteries and heart disease.
Acceding to the EFSA
Moderate intake of phytosterol can lower blood cholesterol level and reduce the risk of coronary heart disease. [12]
The U.S. Food and Drug Administration (FDA) allows food labels to be noted:
Phytosterol can reduce the risk of coronary heart disease. [13]
Health Canada also allows it to be shown on food labels that plant sterol can help reduce cholesterol level. [14]
Although phytosterol has good stability, it can cause oxidation when it is exposed to high temperatures or the air. Eating oxidizing phytosterols can lead to inflammation, unstable hormone levels, carcinogenicity and other health problems.
Application
Industrial grade: it can be used as the starting material for steroid drugs
Food grade: it can be used as food additives, health care raw materials
Specification
Specification
| Items | 90/50 Size | 95/50 Size |
|---|---|---|
| Total sterol | ≥90.0% | ≥95.0% |
| β-sitosterol | ≥50.0% | ≥50.0% |
| Stigmasterol | / | / |
| Campesterol | / | / |
| Residue on ignition | ≤0.5% | ≤0.5% |
| Loss on drying | ≤2.0% | ≤2.0% |
| Heavy metal | ≤10mg/kg | ≤10mg/kg |
| Arsenic | ≤2mg/kg | ≤2mg/kg |
| Solubility | Soluble in thermal ethanol, slightly soluble in ethanol, insoluble in water. | Soluble in thermal ethanol, slightly soluble in ethanol, insoluble in water. |
Packaging
20 kg/drum
Storage
Store in airtight, light-tight and dry condition
Rice bran sterol
CAS number: 83-46-5
HS custom number: 2906199090
National standard code: WS-10001-(HD-0908)-2002
Introduction
Sterol, also known as Rice Bran Sterol Tablets in China, is used for bleeding gums, periodontal abscesses and other conditions caused by periodontal disease.
Specification
| Items | specification |
|---|---|
| Total sterol | ≥63.0% |
| unsaponifiable matter | ≥90.0% |
| Saponification | ≤6.5mg KOH/g |
| Acid value | ≤5.0mg KOH/g |
| Drying on loss | ≤2.0% |
| The total number of aerobic bacteria | ≤1000 cfu/g |
| The total number of molds and yeasts | ≤100 cfu/g |
| E. coli | Negative |
| Appearance | Yellow-brown thick oily, tasteless solid with a little smell of oil |
| Solubility | Soluble in hot ethanol, insoluble in water |
Packaging
25 kg/drum
Storage condition
Prevented from light and stored in airtight and dry condition
Reference
- "The Nobel Prize and the Discovery of Vitamins". www.nobelprize.org.
- Barron, Jon (21 September 2010). "Black Rice Bran, the Next Superfood?". Baseline of Health Foundation.
- S. M. Ghazani; A. G. Marangoni (4 January 2016). "Healthy Fats and Oils". Encyclopedia of Food Grains, Vol: 2-4, 257-267.
- "Vitamin E". Micronutrient Information Center, Linus Pauling Institute, Oregon State University, Corvallis, OR. 1 October 2015.
- "Vitamin E". Office of Dietary Supplements, US National Institutes of Health. 12 July 2019.
- Serbinova E, Kagan V, Han D, Packer L (1991). "Free radical recycling and intramembrane mobility in the antioxidant properties of alpha-tocopherol and alpha-tocotrienol". Free Radical Biology & Medicine. 10 (5): 263–75.
- 吴轲, 孙涵潇,祝 捷,蔡美琴.中国油脂[J].常见食用植物油中维生素 E 异构体含量调查研究,2019,(44-10):95-99.
- Fuji M, Butler JP, et al. Gamma-oryzanol for behavioural and psychological symptoms of dementia. Psychogeriatrics. 2018;18(2):151-152.
- 中华人民共和国卫生部药典委员会.中华人民共和国卫生部药品标准化学药品及制剂[M].谷维素, 1989,(1):H1-55.
- Zduńska K, Dana A, Kolodziejczak A, Rotsztejn H: Antioxidant Properties of Ferulic Acid and Its Possible Application. Skin Pharmacol Physiol 2018;31:332-336.
- Sakai, S.; Kawamata, H.; Kogure, T.; Mantani, N.; Terasawa, K.; Umatake, M.; Ochiai, H. (1999). "Inhibitory effect of ferulic acid and isoferulic acid on the production of macrophage inflammatory protein-2 in response to respiratory syncytial virus infection in RAW264.7 cells". Mediators of Inflammation. 8(3): 173–175.
- European Food Safety Authority. Scientific Opinion of the Panel on Dietetic Products Nutrition and Allergies on a request from Unilever PLC/NV on Plant Sterols and lower/reduced blood cholesterol, reduced the risk of (coronary) heart disease. The EFSA Journal (2008) 781, 1–12.
- U.S. Food & Drug Administration. Health claims: plant sterol/stanol esters and risk of coronary heart disease (CHD). 21 C.F.R. §101.83 (2017, April 1).
- Health Canada. (2010, May 26). Plant Sterols and Blood Cholesterol Lowering.
Ginkgo series
Ginkgo, a tree native to China, stands as one of the world's oldest tree species, dating back to the Carboniferous period, around 230,000 years ago. With an average lifespan ranging from 2000 to 4000 years, ginkgo showcases remarkable tenacity and vitality.
Having served as a traditional Chinese medical material, ginkgo has been cultivated and utilized for thousands of years, highlighting its enduring importance in traditional practices.
-
In 2011, we initiated the trial production of ginkgo biloba extract in our first workshop. Despite the industry's chaotic landscape at that time, we remained steadfast in our commitment to manufacturing authentic products.
-
In 2015, the former CFDA (now the State Administration for Market Regulation) conducted an extensive investigation of 90 ginkgo biloba extract manufacturers nationwide. Delekang stood out as one of the few companies whose products were all certified as qualified. Our unwavering dedication to quality was recognized, and we secured a strong position in the market.
-
In 2016, Delekang's ginkgo biloba extract was approved as an API (Active Pharmaceutical Ingredient) by the former FDA of Jiangsu province. Through collaborations with various drug companies, we continuously optimized the quality of our ginkgo biloba extract, ensuring its stability in various dosage forms.
-
By 2019, we achieved a significant breakthrough in quality. Ginkgolic acid was controlled below 1ppm, and ginkgo toxin below 50 ppm, meeting the EU regulation on maximum pesticide residue levels (EC No 396/2005).
-
In the same year, during the ginkgo biloba extraction process, we discovered shikimic acid, offering a cost-effective solution that reduced its market price. Building on this discovery, we achieved mass production of shikimic acid in 2020, further enhancing the accessibility of this valuable product.
The data speaks for itself. Over the past decade, our technical progress has enabled us to make high-quality products more affordable and accessible to a wider audience.
Ginkgo biloba extract
CAS number:90045-36-6
HS code:13021940.00
Summary
In the 1960s, the Dr. Willmar Schwabe Group developed standardized Ginkgo biloba extract EGb 761, known for promoting vasodilation and enhancing blood flow. This extract is used to treat various conditions, including memory loss and tinnitus[1]. In 2014, the assessment report by EMA (European Medicines Agency) acknowledged the therapeutic benefits of ginkgo biloba extract in improving cognitive impairment associated with aging and enhancing the quality of life in mild dementia[2]. Additionally, Ginkgo biloba extract is widely utilized as a dietary supplement in the United States[3]. In China, the main functions of ginkgo biloba extract preparations are:
Enhancing blood circulation to alleviate obstruction in collaterals, this treatment is suitable for patients experiencing chest blockage, heartache, stroke, hemiplegia, stiff tongue due to blood stasis and collateral blockage, as well as stable angina pectoris and cerebral infarction associated with the mentioned syndromes.[4]
Delekang's Ginkgo biloba extract has received approval from the State Administration for Market Regulation (previously known as the Food and Drug Administration) and is authorized for use as an API (Active Pharmaceutical Ingredient) in the production of diverse drug preparations. Additionally, we have obtained the "Food Production License", allowing us to manufacture Ginkgo biloba extract for dietary supplements.
Our Advantages
-
Ginkgo biloba extract is a commonly used ingredient produced by many plant extract manufacturers. However, achieving good, let alone excellent, quality is a challenge that only a few can accomplish. Over the past decade, we have continuously elevated the quality of our ginkgo biloba extract through the following measures:
-
We have established long-term partnerships with ginkgo leaf suppliers, enabling us to collect and analyze extensive quality data from various regions. This helps us identify suitable raw materials for standardized products with consistent quality.
-
Our production process adheres to the standards set by Chinese Pharmacopoeia, U.S. Pharmacopoeia, and European Pharmacopoeia.
-
We comply with the EU regulations on maximum pesticide residue levels (EC No 396/2005).
-
Our extract contains ginkgolic acid below 1ppm and ginkgotoxin below 50ppm.
-
Our production line has received approval from China FDA.
-
Our extract is compatible with most oral dosage forms, ensuring ease of use for various pharmaceutical applications.
-
-
In addition to ensuring high quality, Delekang strives to make its products more affordable. As a specialized manufacturer, we have the following advantages:
-
We have developed advanced technology that allows us to fully utilize raw materials, reducing the cost of production. Extracted ginkgo leaves and waste materials can be repurposed to extract sodium copper chlorophyllin and shikimic acid, while used leaves serve as fuel, providing renewable energy for the production line.
-
Our scale of operation helps dilute fixed costs per unit, including investments in research and development, equipment, and facilities, leading to more cost-effective production.
-
Unlike generic extraction equipment, our specialized production line designed for ginkgo biloba enables more effective quality control and cost reduction during production.
-
Specifications
| Items | CP | USP | EP | In-house |
|---|---|---|---|---|
| Flavonol glycosides | ≥24.0% | 22.0-27.0% | 22.0-27.0% | 22.0-27.0% |
| Terpene lactones | ≥6.0% | 5.4-12.0% | 5.4-6.6% | 5.4-12.0% |
| Bilobalide | / | 2.6-5.8% | 2.6-3.2% | 2.6-5.8% |
| Ginkgolide | / | 2.8-6.2% | 2.8-3.4% | 2.8-6.2% |
| Quercetin/kaempferol ratio | 0.8-1.2 | / | / | 0.8-1.2 |
| Isorhamnetin/Quercetin ratio | ≥0.15 | / | / | ≥0.15 |
| Ginkgolic acid | ≤5mg/kg | ≤5mg/kg | ≤5mg/ kg | ≤1mg/kg |
| Free quercetin | ≤10mg/g | ≤5mg/g | / | ≤10mg/g |
| Free kaempferol | ≤10mg/g | / | / | ≤10mg/g |
| Free isorhamnetin | ≤4mg/g | / | / | ≤4mg/g |
| Rutin | / | ≤4.0% | / | / |
| Pesticide residues | / | USP<561> | EC396/2005 | / |
| Loss on drying | ≤5.0% | ≤5.0% | ≤5.0% | ≤4.0% |
| Residue on ignition | ≤0.8% | / | / | ≤0.8% |
| Heavy metals | ≤20m g/kg | / | / | ≤15m g/kg |
| HPLC Fingerprint | ≥0.90 | / | / | ≥0.90 |
Package and storage
25 kg/drums; protected from light; stored in airtight and dry condition
Shikimic acid
CAS number: 138-59-0
HS Custom code: 2918190090
Molecular mass: 174.15
Structure
Specification
| Items | sepcifications |
|---|---|
| Specific rotation[α]D 25°C/° | -175~-184 |
| Content | ≧98.0% |
| Heavy metals | ≦20ppm |
| Residue on ignition | ≤0.8 % |
| Loss on drying | ≤1.0% |
| Solubility | 18% m/v (soluble in water) |
| Appearance | white to light yellow fine powder |
| Plant origin | Ginkgo biloba L. |
Packaging and storage
25kg/drum Protect from light and stored in airtight and dry condition
Quinic acid
CAS编号:77-95-2
Chemical formula:C7H12O6
Molecular mass:192.17
Structure
Specification
| Items | sepcifications |
|---|---|
| Specific rotation[α]D 25°C/° | -42~-44 |
| Content | ≧98.0% |
| Heavy metals | ≦20ppm |
| Residue on ignition | ≤0.8 % |
| Loss on drying | ≤1.0% |
| Appearance | white to light yellow crystalline powder |
| Plant origin | Ginkgo biloba L. |
Packaging and storage
25kg/drum Protect from light and stored in airtight and dry condition
References
- EGb 761: ginkgo biloba extract, Ginkor. Drugs R D. 2003;4(3):188-93. doi: 10.2165/00126839-200304030-00009. PMID: 12757407.
- Committee on Herbal Medicinal Products. "Assessment report on Ginkgo biloba L., folium" (PDF). European Medicines Agency.
- Z. Pang, F. Pan, and S. He.The Journal of Alternative and Complementary Medicine.Sep 1996.359-363.
- Chinese Pharmacopoeia Commission. Pharmacopoeia of the People's Republic of China[M].Ginkgo Leaves Extract, 2020,(1):1614.
Green Tea series
Green tea extract
CAS number: 84650-60-2
HS customs number:
Appearance
Tea polyphenols, freely soluble in water, ethanol, ethyl acetate and slightly soluble in oils, are white to brown powdery solids or crystals with astringent taste. They are stable under hot or acidic condition in the range of PH 2~7. They are slightly hygroscopic, the PH of their aqueous solution is 3~4. Tea polyphenols are easy to be oxidized and become brown under alkaline condition. Green-black compounds are formed when they meet iron ions.
Specification
| Specifications | 98/80/50 | 95/75/45 | 90/70/40 | TP |
|---|---|---|---|---|
| Total polyphenols | ≥98.0% | ≥95.0% | ≥90.0% | 50/60/70/80% |
| Catechins | ≥80.0% | ≥75.0% | ≥70.0% | / |
| EGCG | ≥50.0% | ≥45.0% | ≥40.0% | / |
| Caffeine | ≤0.5% | ≤1.0% | ≤1.0% | ≤2.0% |
| Loss on drying | ≤5.0% | ≤5.0% | ≤5.0% | ≤5.0% |
| Residue on ignition | ≤3.0% | ≤3.0% | ≤3.0% | ≤3.0% |
| Heavy metals | ≤10.0 mg/kg | ≤10.0 mg/kg | ≤10.0 mg/kg | ≤10.0 mg/kg |
| Lead | ≤5.0 mg/kg | ≤5.0 mg/kg | ≤5.0 mg/kg | ≤5.0 mg/kg |
| Arsenic | ≤2.0 mg/kg | ≤2.0 mg/kg | ≤2.0 mg/kg | ≤2.0 mg/kg |
Packaging
25kg/drum
Storage condition
Store in dark, airtight and dry place.
L-Theanine
CAS number: 3081-61-6
HS customs number:
Structure
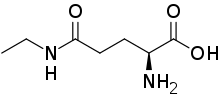
Quality specification
| Specification | TP20 | TP30 | TP40 | TP90 |
|---|---|---|---|---|
| Appearance | light yellow to light brown or dark brown powder | white or almost white powder | ||
| Loss on drying | ≤6.0% | |||
| Heavy metal | ≤10mg/kg | |||
| Lead | ≤2mg/kg | |||
| Arsenic | ≤2mg/kg | |||
| Theanine | ≥20.0% | ≥30.0% | ≥40.0% | ≥90.0% |
| Total aerobic bacteria(cfu/g) | ≤1000 | |||
| Total molds and yeasts(cfu/g) | ≤100 | |||
| Escherichia Coli(1g) | negative | |||
Package
20kg/drum
Storage condition
Protect from light and stored in airtight and dry condition


